 
Explain Discuss the conditions needed for covalent bonding and the stable molecule that is formed.In a covalent bond, electrons from each atom are attracted or “shared” by both atoms. This type of bond is called a covalent bond. At the end of the animation, explain that the individual hydrogen atoms have now bonded to become the molecule H 2. But the attractions are strong enough to pull the two atoms close enough together so that the electrons feel the attraction from both protons and are shared by both atoms. Point out to students that the attractions are not strong enough to pull the electron completely away from its own proton. Explain that if the atoms get close enough to each other, the electron from each hydrogen atom feels the attraction from the proton of the other hydrogen atom (shown by the double-headed arrow). Remind students that the electron and its own proton are attracted to each other. Make sure students see that each hydrogen atom has 1 proton and 1 electron. Project the animation Covalent bond in hydrogen. Introduce the question students will investigate in this lesson: If atoms have an equal number of protons and electrons, why do atoms bond to other atoms? Why don’t they just stay separate? Begin to answer this question by using hydrogen as an example. 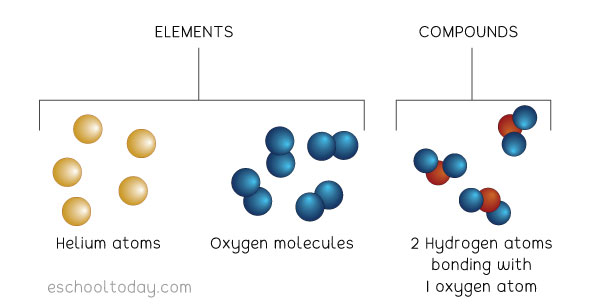
Engage Show an animation to introduce the process of covalent bonding.This lesson will probably take more than one class period. 2 wires with alligator clips on both ends.Safetyīe sure you and the students wear properly fitting goggles. The activity sheet will serve as the “Evaluate” component of each 5-E lesson plan. Evaluationĭownload the student activity sheet, and distribute one per student when specified in the activity. Students will be able to draw a model of the covalent bonds between the atoms in H 2 (hydrogen), H 2O (water), O 2 (oxygen), CH 4 (methane), and CO 2 (carbon dioxide). Students will be able to explain that attraction between the protons and electrons of two atoms cause them to bond. Students will consider why atoms bond to form molecules like H 2 (hydrogen), H 2O (water), O 2 (oxygen), CH 4 (methane), and CO 2 (carbon dioxide).  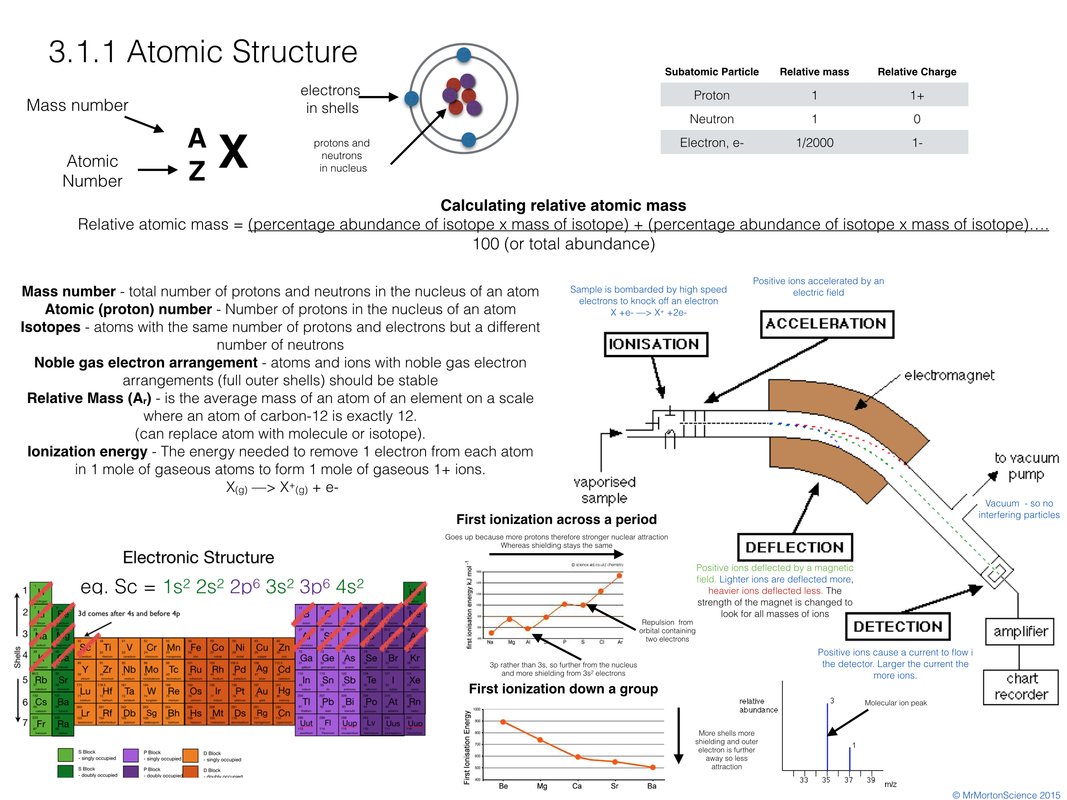
Students will look at animations and refer to the energy level models they have been using to make drawings of the process of covalent bonding.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
